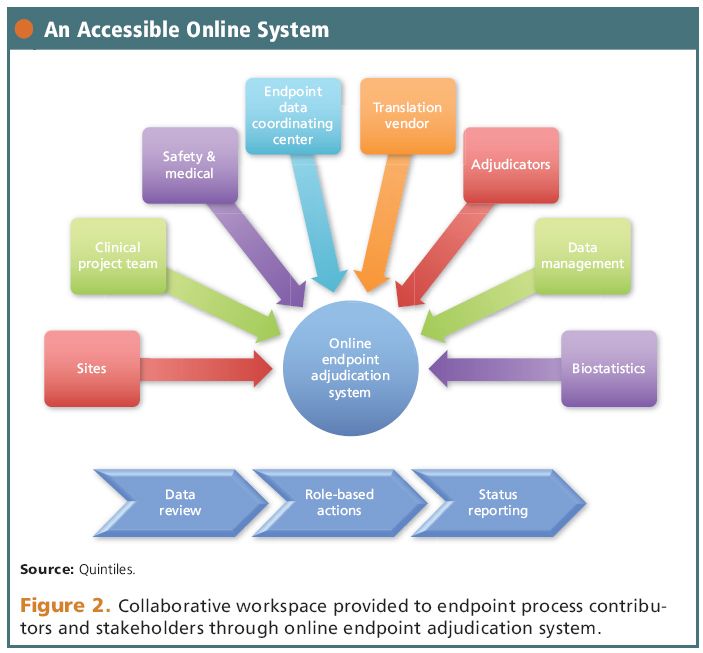
A polymer-coated, paclitaxel-eluting stent (Eluvia) versus a polymer-free, paclitaxel-coated stent (Zilver PTX) for endovascular femoropopliteal intervention (IMPERIAL): a randomised, non-inferiority trial - The Lancet

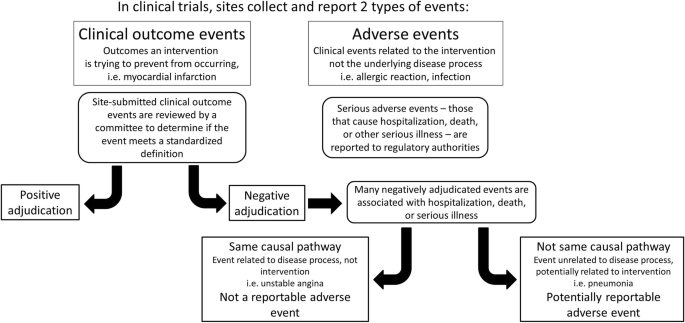
Clinical events classification (CEC) in clinical trials: Report on the current landscape and future directions — proceedings from the CEC Summit 2018 - ScienceDirect

GLASSY design. CEC, Clinical Event Committee; GLASSY, GLOBAL LEADERS... | Download Scientific Diagram

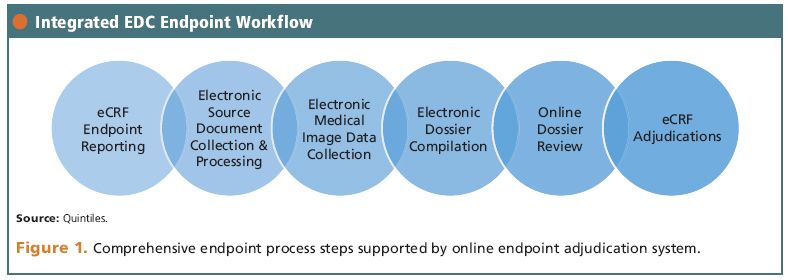
PDF) Clinical Trials: Minimising source data queries to streamline endpoint adjudication in a large multi-national trial

Clinical events classification (CEC) in clinical trials: Report on the current landscape and future directions — proceedings from the CEC Summit 2018 - ScienceDirect

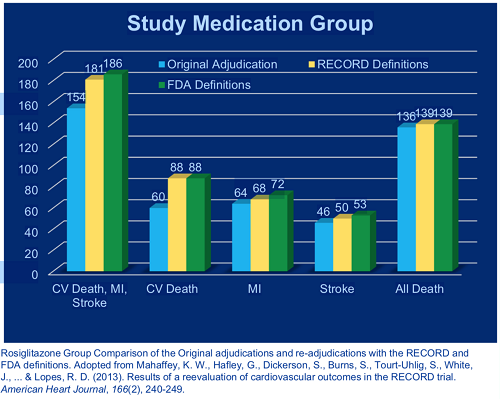
Central Adjudication Identified Additional and Prognostically Important Myocardial Infarctions in Patients Undergoing Percutaneous Coronary Intervention | Circulation: Cardiovascular Interventions

Clinical endpoint adjudication in a contemporary all-comers coronary stent investigation: Methodology and external validation - ScienceDirect

Advanced Analytics is revolutionizing clinical trials: the experience of Exom Group, the first European digital CRO, to implement such an innovative approach. | Exom Group