Beyond Celiac on Twitter: "RESEARCH NEWS: Nexvax2 vaccine protects against high doses of gluten, study shows. Read more about the Nexvax2 vaccine here: https://t.co/A8eqESeLVJ #celiac #celiacresearch #Nexvax2 @ImmusanT" / Twitter

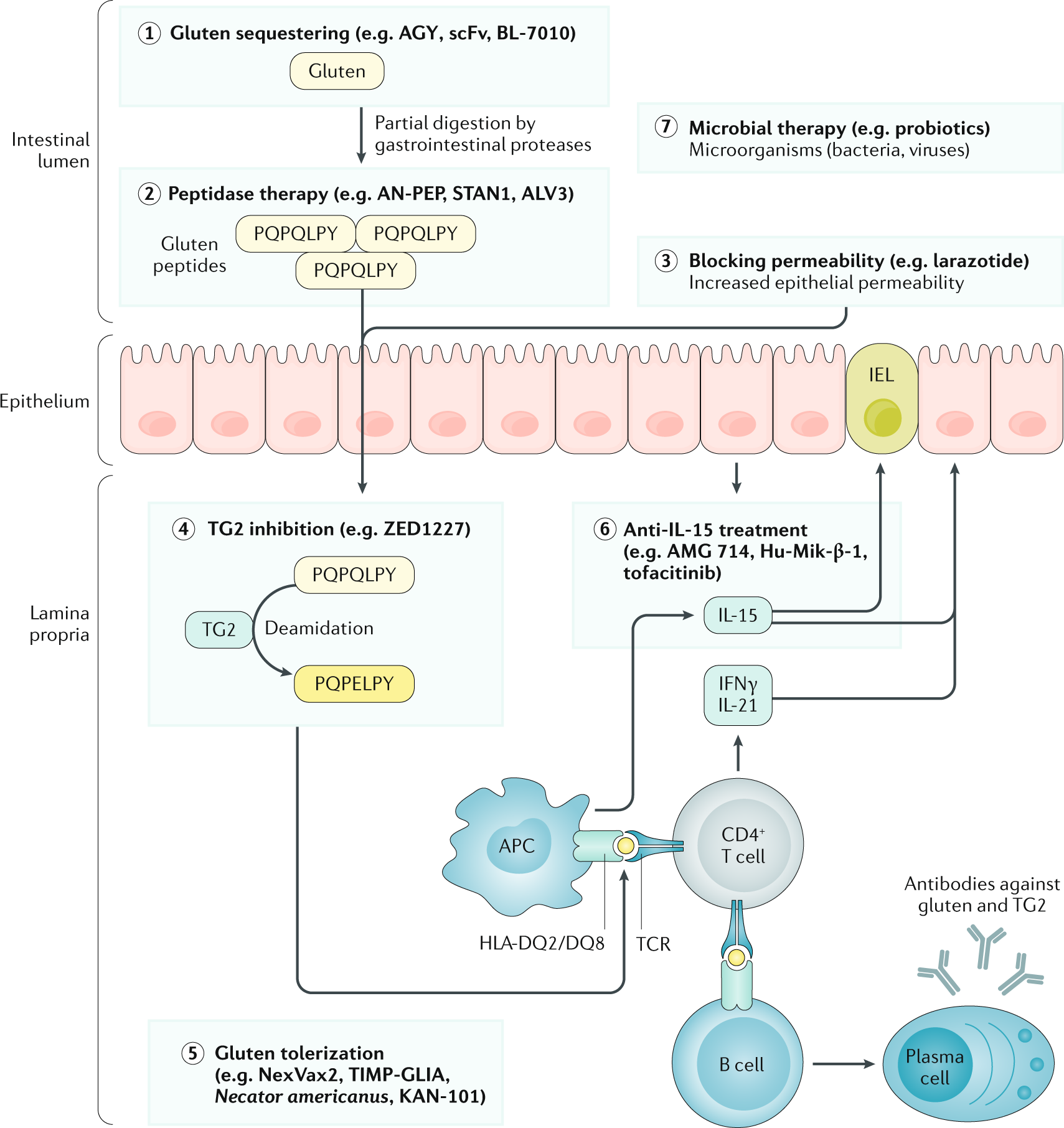
Randomised clinical trial: a placebo‐controlled study of subcutaneous or intradermal NEXVAX2, an investigational immunomodulatory peptide therapy for coeliac disease - Truitt - 2019 - Alimentary Pharmacology & Therapeutics - Wiley Online Library

Frontiers | A Sensitive Whole Blood Assay Detects Antigen-Stimulated Cytokine Release From CD4+ T Cells and Facilitates Immunomonitoring in a Phase 2 Clinical Trial of Nexvax2 in Coeliac Disease | Immunology

A Participant's Perspective on the Failure of the Nexvax2 "Celiac Disease Vaccine" Clinical Trial - Celiac.com

Randomised clinical trial: a placebo‐controlled study of subcutaneous or intradermal NEXVAX2, an investigational immunomodulatory peptide therapy for coeliac disease - Truitt - 2019 - Alimentary Pharmacology & Therapeutics - Wiley Online Library

ImmusanT Discontinues Phase 2 Clinical Trial for Nexvax2® in Patients With Coeliac Disease - Wesley Medical Research

Randomised clinical trial: a placebo‐controlled study of subcutaneous or intradermal NEXVAX2, an investigational immunomodulatory peptide therapy for coeliac disease - Truitt - 2019 - Alimentary Pharmacology & Therapeutics - Wiley Online Library

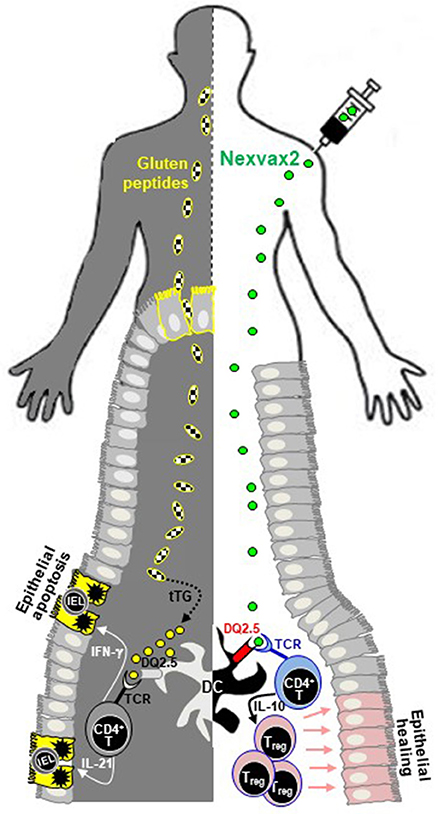
ImmusanT Publishes Positive Data from Phase 1 Trials of its Celiac Disease Drug Nexvax2 - Drug Discovery and Development